
What are the fees for registering Saudi Saber certification?
All goods imported into Saudi Arabia need to be registered on Saber, followed by applications for PC product certificates and SC customs clearance certificates. The cost of applying for these certificates is also a major concern for everyone.
What are the components of the fees for Saber certification registration?
The fees for Saber certification vary depending on factors such as product category, risk level, testing scope, and duration. Some products require factory inspection, which increases costs.
Below are the fees for Saber certification:
Application Fee:
The application fee varies depending on the product category, generally ranging between $800 and $1000 USD.
Registration Fee:
The registration fee is determined based on the product's risk level and category, typically ranging from $100 to $400 USD.
Annual Supervision Inspection Fee:
This fee is determined based on the product category and risk level, requiring an annual payment of a certain amount. Specific amounts vary depending on the product, generally ranging in the hundreds of dollars.
On-Site Audit Fee:
The cost is calculated based on the audit scope and duration, typically charged per day. This fee varies depending on the product type and complexity of the audit.
Product Testing Fee:
This fee depends on the actual product and varies. Laboratories generally accept IEC reports; if reports are unavailable, testing must be conducted. Testing fees for simple toys may start at $700 to $1000 USD, whereas testing fees for large mechanical products could range from $6000 to $8000 USD.
Factory Inspection Cost:
Fees charged by different agencies vary, generally ranging from $600 to $700 USD. Products requiring factory inspection, such as building materials, tableware, automotive parts, and amusement equipment, incur additional factory inspection fees.
Certificate Review Fee:
The certificate review fee for each product is approximately $650 USD.
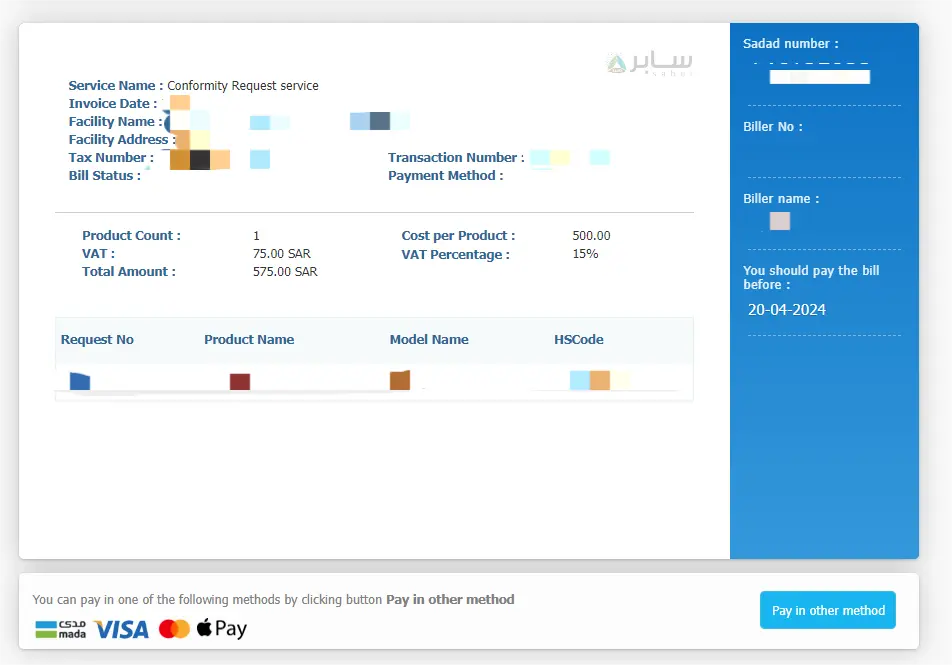
Saber Platform Registration Fee:
Platform registration fees can be paid directly on the platform:
- PC certificate: 575 Saudi Riyals
- SC certificate: 455 Saudi Riyals
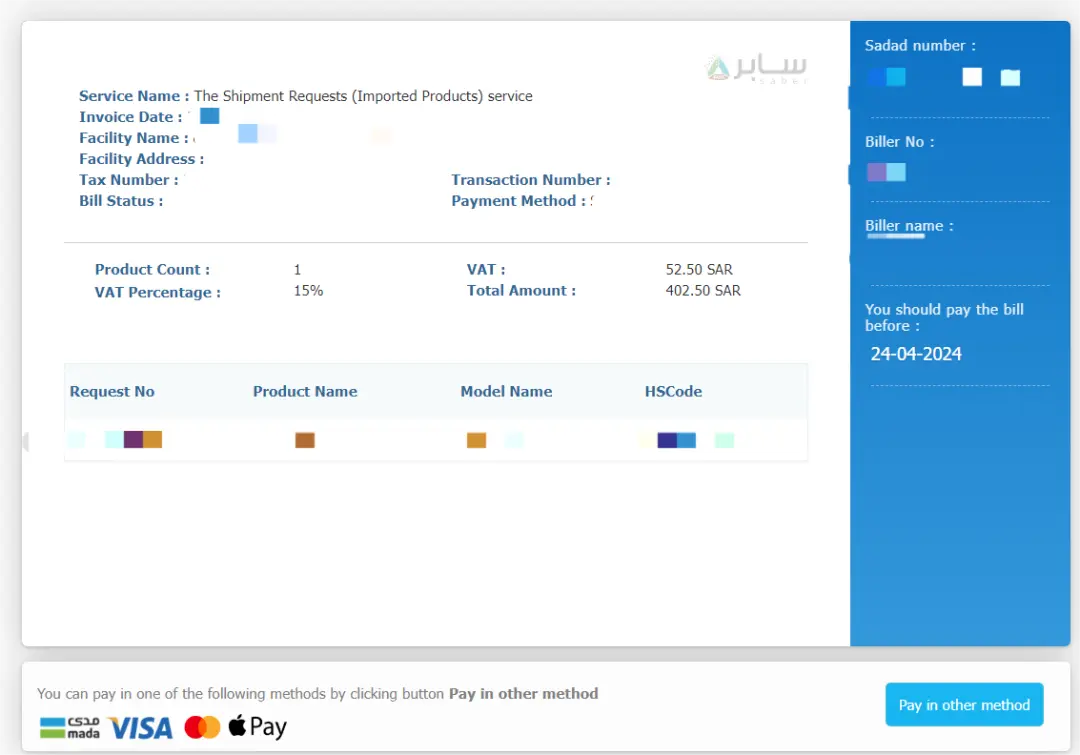
In summary, actual costs may vary based on the product's risk level, chosen certification body, and specific service requirements. The final cost should be based on the actual quotation provided.
JJR Laboratory in China holds ISO 17025 accreditation and offers Saber certification testing services. We are located in China and can help you save 30% on certification testing fees."
Email:hello@jjrlab.com
Write your message here and send it to us
 Mobile DVR CE Testing Lab
Mobile DVR CE Testing Lab
 Telematics Device CE Compliance Standards
Telematics Device CE Compliance Standards
 CE Certification Requirements for Electronic Toys
CE Certification Requirements for Electronic Toys
 CE Testing Services for Electronics
CE Testing Services for Electronics
 CE Compliance Testing Lab
CE Compliance Testing Lab
 Toy Safety Testing Australia
Toy Safety Testing Australia
 Canada Toy Testing
Canada Toy Testing
 Toy Safety Testing USA
Toy Safety Testing USA
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




