
Button Battery UL 4200A-2023 Testing Laboratory
The regulation governing button batteries or coin cells and consumer products containing such batteries in the United States includes 16 cfr part 1263 and ANSI/UL 4200A-2023 standards, which officially came into effect on March 19, 2024. China’s JJR Laboratory possesses testing capabilities for 16 cfr 1263 and has obtained CNAS accreditation while also being recognized by the U.S. CPSC laboratory. Additionally, JJR Laboratory is an authorized partner laboratory for the Amazon platform, assisting businesses in issuing CPC and gcc certificates to confirm product compliance.

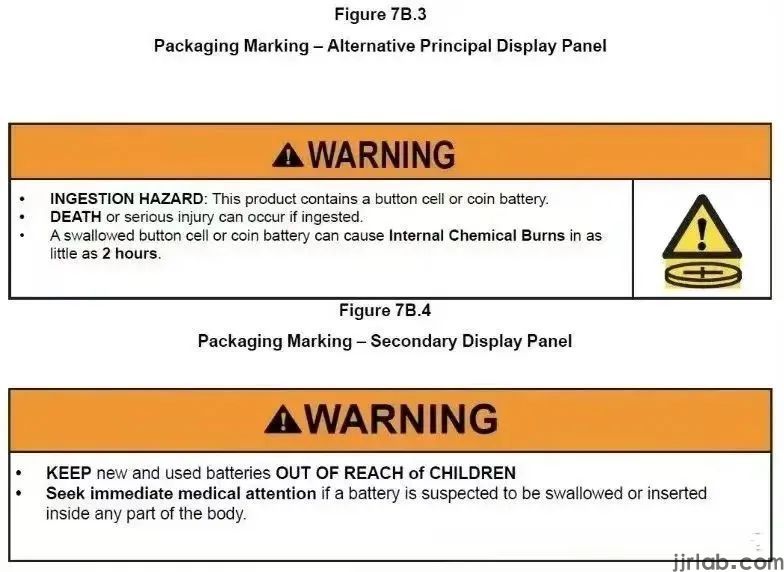
The requirements of 16 CFR Part 1263 and ANSI/UL 4200a-2023 for consumer products containing button batteries or coin cells are as follows:
1. Battery compartments containing replaceable button batteries or coin cells must be secured in a manner that requires tools or at least two independent actions to open.
2. Button batteries or coin cells must not be accessible to children due to normal use or misuse testing.
3. Product packaging must include warning labels.
4. If applicable, the product itself must carry warning labels.
5. The accompanying instruction manual must include all relevant warnings.
Friendly Reminder
The mandatory regulations of 16 CFR Part 1263 significantly impact all consumer products containing button batteries or coin cells, including children’s products, as the regulation mandates third-party laboratory testing by the CPSC. JJR Laboratory in China advises sellers on Amazon U.S. to comply with Amazon and relevant laws and regulations, and to promptly upload product testing reports on the Amazon seller platform while paying attention to related requirements to avoid the risk of product delisting.
Email:hello@jjrlab.com
Write your message here and send it to us
 What Are the Safety Tests for Lithium Batteries?
What Are the Safety Tests for Lithium Batteries?
 Wireless Device EMC Certification
Wireless Device EMC Certification
 Type-C Interface EN62680-1 Testing
Type-C Interface EN62680-1 Testing
 Electrical Safety Test IEC 60601
Electrical Safety Test IEC 60601
 IEC 60601-1-2 EMC Testing
IEC 60601-1-2 EMC Testing
 Polycyclic Aromatic Hydrocarbons (PAHs) Testing La
Polycyclic Aromatic Hydrocarbons (PAHs) Testing La
 EU EN71 Certification Testing for Mechanical Toys
EU EN71 Certification Testing for Mechanical Toys
 EU Toy Safety Testing (EN 71 / 2009/48/EC)
EU Toy Safety Testing (EN 71 / 2009/48/EC)
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




