
U.S. CPSC to Launch eFiling for Compliance Certificates
The U.S. Consumer Product Safety Commission (CPSC) has issued a Supplemental Notice of Proposed Rulemaking (SNPR) to amend the compliance certificate regulations under 16 CFR 1110.
This SNPR proposes aligning the certificate rules with other CPSC rules on testing and certification, while collaborating with U.S. Customs and Border Protection (CBP) to streamline the submission process for consumer product compliance certificates through electronic filing (eFiling). This includes Children's Product Certificates (CPC) and General Certificates of Conformity (GCC).
1. Children’s Product Certificate (CPC)– A declaration by manufacturers or importers that their children’s products comply with all applicable children’s product safety rules (bans, standards, similar rules or regulations).
2. General Certificate of Conformity (GCC)– A declaration by manufacturers or importers that general use products (non-children’s products) comply with all applicable consumer product safety rules.
Consumer product compliance certificates are critical documents that verify a product meets safety standards and must accompany goods entering the U.S. market. The core of the eFiling program is to simplify the submission of these certificates by leveraging digital tools for more efficient, accurate, and timely collection of compliance data. Through eFiling, the CPSC can better assess consumer product risks and quickly identify non-compliant goods. This not only helps intercept non-compliant products at ports but also accelerates the entry of compliant products into the market.
Currently, the CPSC is actively developing and finalizing rules for eFiling. The program is expected to be formally implemented and become mandatory as early as 2025. Going forward, importers will be required to electronically file data elements from certificates that comply with CBP requirements.
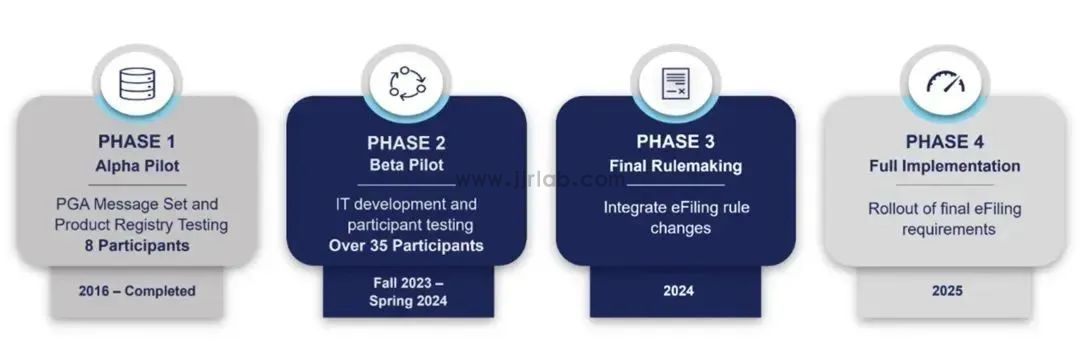
When submitting via CPSC eFiling, importers should provide at least the following seven categories of data:
3. Finished product identification (e.g., Global Trade Item Number (GTIN) data).
4. Each consumer product safety rule for which the product is certified.
5. Product production date.
6. Product manufacturing, production, or assembly location, including the manufacturer’s name, full address, and contact information.
7. Date of the most recent test demonstrating compliance with the applicable consumer product safety rules.
8. Information about the testing laboratory relied upon for the certificate, including the lab’s name, full address, and contact information.
9. Contact information of the individual maintaining the test records, including name, full address, and contact details.
What are the benefits of implementing eFiling?
1. For traditional certificate filing, eFiling simplifies the submission process, enabling the CPSC to better assess product risks, quickly identify non-compliant products, and efficiently manage systematic compliance.
2. For importers of regulated consumer products, it accelerates the smooth entry of compliant products into the market, improving trade efficiency.
3. For consumers, it ensures safer consumer products enter the market, benefiting public safety with no downside.
Email:hello@jjrlab.com
Write your message here and send it to us
 US Customs Regulations: Latest IOR Rules
US Customs Regulations: Latest IOR Rules
 Trump Signs Executive Order: New U.S. IOR Regulati
Trump Signs Executive Order: New U.S. IOR Regulati
 EU Compliance for Exporting Pacifier Clips
EU Compliance for Exporting Pacifier Clips
 Lighting Fixtures Australian SAA Certification
Lighting Fixtures Australian SAA Certification
 Food Contact Material (FCM) Compliance Testing
Food Contact Material (FCM) Compliance Testing
 Children's Jewelry Amazon Compliance Services
Children's Jewelry Amazon Compliance Services
 Amazon Children's Plush Toys Testing and Complianc
Amazon Children's Plush Toys Testing and Complianc
 Guide to the US Children's Product CPC Certificati
Guide to the US Children's Product CPC Certificati
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




