
Do Class I Medical Devices Need an MDR?
Class I medical devices placed on the market under the MDD Directive and still classified as Class I devices under the MDR Regulation must comply with the requirements listed in the MDR Regulation starting from May 26, 2021.

Definition of Class I Medical Devices:
Class I medical devices, according to the ISO 14971 standard, are defined as medical devices with potential low risk due to the following reasons:
1. Short or limited contact time with the human body
2. For external use or causing minimal impact on the human body
3. Do not contain active components (e.g., batteries or cables)
4. Not intended for implantation or penetration into the human body (non-sterile products)
Examples of Class I Medical Devices:
1. Medical dressings and bandages
2. Dental instruments (e.g., toothbrushes, mouthwash)
3. Hearing aids
4. Assistive devices such as wheelchairs and crutches
CE certification MDR Medical Device Directive:
The CE Certification MDR refers to the Medical Device Regulation (MDR) 2017/745, an EU regulation that came into effect on May 26, 2021. The MDR replaces the previous Medical Devices Directive (MDD) 93/42/EEC.
CE Certification for Class I Medical Devices:
- Sign an authorized representative (EU Rep) agreement
- Prepare technical documentation and application materials
- Register the product with the EU medical device regulatory authority
- Issue a Declaration of Conformity (DOC)
ce certification process for Class I Medical Devices:
1. Determine Applicable Regulations:
Class I medical devices are regulated under the Medical Device Regulation (MDR) 2017/745.
2. Classify the Medical Device:
Manufacturers need to classify the medical device as Class I according to the rules in Annex VIII of the MDR.
3. Meet Basic Requirements:
Manufacturers must ensure that the medical device meets the basic requirements specified in Annex I of the MDR.
4. Technical Documentation:
Manufacturers need to compile technical documentation, including information on the design, manufacturing, and performance of the medical device.
5. Declaration of Conformity:
Manufacturers must draft and sign a Declaration of Conformity as per Annex III of the MDR.
6. Self-Declaration of ce mark:
Manufacturers can affix the CE mark on the medical device, indicating that it meets the basic requirements and has obtained CE certification.
Certification Documentation Timeline:
- Complete the application form, provide product instructions, and production process flow.
- The process takes approximately 2 to 4 weeks.
Note:
Most Class I devices do not require product testing. The MDR came into effect on May 26, 2021, and will fully replace the MDD on May 26, 2024. Specific compliance requirements may vary based on the function and type of the medical device. Manufacturers are advised to contact China's JJR Laboratory for the latest information and guidance. We can help you save 30% on certification costs.
Email:hello@jjrlab.com
Write your message here and send it to us
 Amazon Clothing Storage Units ASTM F2057-23 and DV
Amazon Clothing Storage Units ASTM F2057-23 and DV
 What is the ASTM F2057 Compliance Standard?
What is the ASTM F2057 Compliance Standard?
 US Dresser ASTM F2057-23 Furniture Tip Over Test
US Dresser ASTM F2057-23 Furniture Tip Over Test
 The Latest 2026 Thailand NBTC Compliance Guide
The Latest 2026 Thailand NBTC Compliance Guide
 CE Certification Directives for Electronic Product
CE Certification Directives for Electronic Product
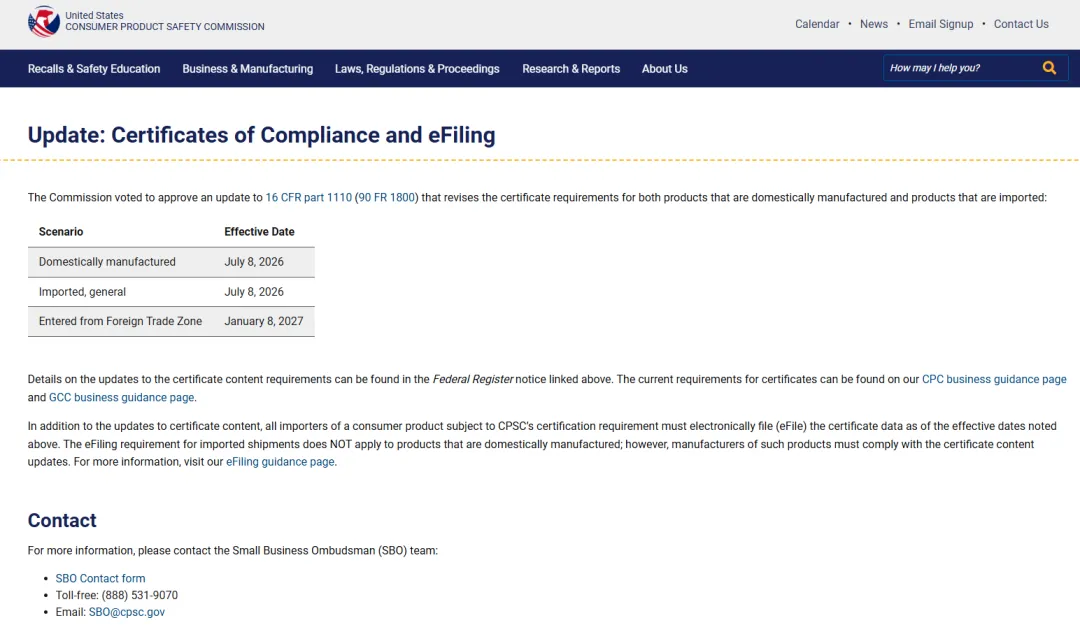
 What is CPSC eFiling
What is CPSC eFiling
 CPSC Products Must Undergo eFiling
CPSC Products Must Undergo eFiling
 What is the U.S. CPSC eFiling System?
What is the U.S. CPSC eFiling System?
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




