
How to Identify the Authenticity of CE Certificates?
When exporting to the European Union market, CE certification is essential. Currently, various types of medical ce certificates have emerged in the market, which can be overwhelming. To better help you verify the authenticity of CE certificates, we will discuss specific methods for identification below.

Checking the Official Website of the Notified Body
Generally, large EU certification bodies will have a certificate inquiry window available on their official website. After logging into the body’s website, there will be a certificate inquiry page.
You can input the manufacturer’s name in English, certificate number, or other relevant details to check if there is a matching CE certificate. If one appears, it suggests the certificate might be authentic.
Of course, this method is only applicable if the issuing body provides a certificate query service. For bodies that do not offer such a service, this method will not work.
So, what if we obtain a medical CE certificate, but the issuing body does not provide an online query service?
Checking the Official EU Website for Notified Bodies
You can also attempt to check the issuing body of the certificate on the official EU website.
For example, when dealing with medical devices, check if the issuing body is authorized under the EU Medical Devices Directive MDD 93/42/EEC or the MDR Medical Device Regulation (EU) 2017/745.
Here is the link to the EU official MDD 93/42/EEC Medical Device Directive authorized body list:
https://ec.europa.eu/growth/toolsdatabases/nando/index.cfmfuseaction=directive.notifiedbody&dir_id=13
From the EU website, you can see that there are 56 authorized notified bodies under the MDD 93/42/EEC Medical Devices Directive. The list of specific bodies, notification numbers, and the range of products they are qualified to certify is detailed.
Starting from May 26, 2020, the MDR (EU) 2017/745 Medical Devices Regulation officially replaced the MDD directive and is now mandatory. Currently, there are only 12 notified bodies authorized under MDR.
Here is the link to the EU official MDR (EU) 2017/745 Medical Device Regulation authorized body list:
https://ec.europa.eu/growth/toolsdatabases/nando/index.cfmfuseaction=directive.notifiedbody&dir_id=34
If the issuing body of your medical CE certificate is not listed in the above databases, it means that the body is not authorized to issue CE certificates for medical products in the EU.
Unfortunately, the CE certificate you have obtained would be invalid.
Additionally, we can analyze the certification process for medical devices to assist in identification.
Analyzing the ce certification process for Identification
For example, with face masks, first confirm whether the mask is classified as a medical device. Masks are divided into medical masks and protective masks, both of which must meet EU regulations when exporting to Europe and the U.S.:
Personal Protective Masks:
- Regulation: EU 2016/425 (PPE)
- Standard: EN 149
Medical Masks:
- Regulation: 93/42/EEC (MDD) or EU 2017/745 (MDR)
- Standard: EN 14683
Personal protective masks are not classified as medical devices, so they do not need to meet EU medical device regulations. They only need to comply with PPE (Personal Protective Equipment) regulations for CE certification. Currently, there are 112 authorized notified bodies under the EU PPE regulation.
You can check the list of authorized bodies under the EU (EU) 2016/425 Personal Protective Equipment regulation here:
https://ec.europa.eu/growth/toolsdatabases/nando/index.cfmfuseaction=directive.notifiedbody&dir_id=155501
Summary:
1. Verify the certificate by checking the related information on the issuing body’s website.
2. Use the EU official websites to verify the issuing body and distinguish authenticity, but note that this method only works for Notified Bodies (NB), as non-NB bodies are not authorized third-party organizations and may not be verifiable.
3. Analyze the certification process to determine whether the issuing body is legitimate (CE certification processes are based on EU regulations, and you need to verify the correct process on the relevant organization’s website).
Email:hello@jjrlab.com
Write your message here and send it to us
 Amazon Clothing Storage Units ASTM F2057-23 and DV
Amazon Clothing Storage Units ASTM F2057-23 and DV
 What is the ASTM F2057 Compliance Standard?
What is the ASTM F2057 Compliance Standard?
 US Dresser ASTM F2057-23 Furniture Tip Over Test
US Dresser ASTM F2057-23 Furniture Tip Over Test
 The Latest 2026 Thailand NBTC Compliance Guide
The Latest 2026 Thailand NBTC Compliance Guide
 CE Certification Directives for Electronic Product
CE Certification Directives for Electronic Product
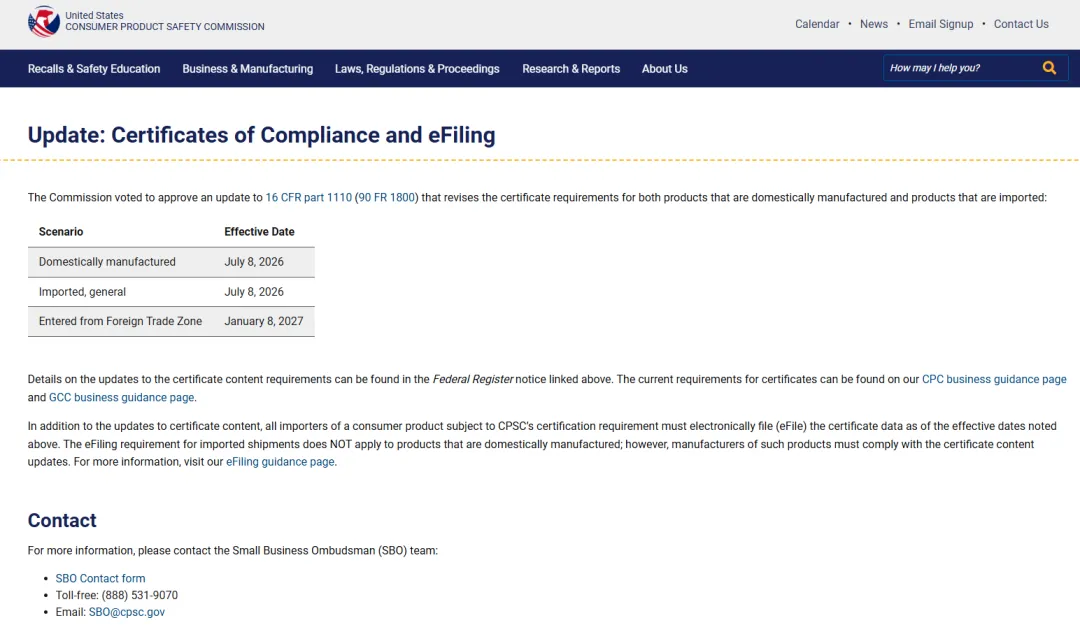
 What is CPSC eFiling
What is CPSC eFiling
 CPSC Products Must Undergo eFiling
CPSC Products Must Undergo eFiling
 What is the U.S. CPSC eFiling System?
What is the U.S. CPSC eFiling System?
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




