
Reusable Medical Device Cleaning Sterilization Verification
Medical device registration (NMPA) declaration requires the submission of cleaning and disinfection sterilization validation as research information.
In the past, this part was basically verified and provided by the enterprises themselves, which were limited by the test equipment, experimental environment, and lack of professional staff to study the regulations and standards. Often the research information provided was not perfect and even failed to pass the verification, which would significantly improve the probability of cross-infection brought by the products in the actual application in hospitals.
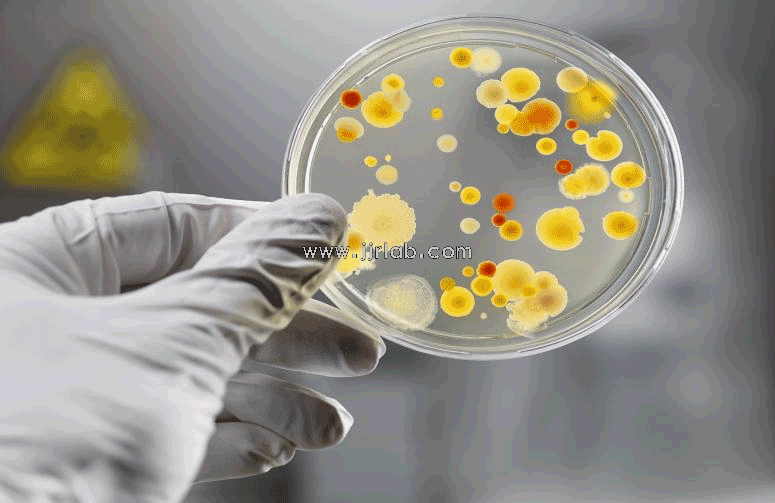
JJR Medical Device Service - Reusable Medical Device Cleaning Disinfection Sterilization Validation Solution successfully reduces the risk of medical device manufacturers and protects people's life safety.
JJR Medical Device Services
Quality service leader in the medical device industry
More than 20 years of experience in the medical device industry
Together with manufacturers, technical experts, and evaluation teachers of local drug regulatory bureaus, we have conducted in-depth research on the use of hospital departments and put forward a series of cleaning and sterilization validation programs for reusable medical devices.
So far, we have completed the validation service for 245 products



Email:hello@jjrlab.com
Write your message here and send it to us
 ROHS and Weee Compliance
ROHS and Weee Compliance
 Waste of Electrical and Electronic Equipment Weee
Waste of Electrical and Electronic Equipment Weee
 Low Voltage Directive CE Marking
Low Voltage Directive CE Marking
 What are CE EMC Testing Requirements
What are CE EMC Testing Requirements
 What are the CE EMC standards
What are the CE EMC standards
 RF Testing Companies
RF Testing Companies
 European WEEE Directive
European WEEE Directive
 European Authorised Representative
European Authorised Representative
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




