
2026 EU SVHC Candidate List (253 Substances)
On 4 February 2026, the European Chemicals Agency (ECHA) completed the assessment of the 36th batch of substances for the EU Candidate List of Substances of Very High Concern (svhc). Following this adjustment, the total number of substances officially included in the list stands at 253, which directly impacts the compliance and market access of all export-to-EU product categories, including electrical and electronic products, consumer goods, chemical products and others.

Overview of Key Adjustments to the List
This update is based on the final assessment of 3 substances under evaluation, with no new entries added for evaluation. The specific changes are as follows:
• Newly added substances: n-Hexane, 4,4'-[2,2,2-trifluoro-1-(trifluoromethyl)ethylidene]BISphenol (BPAF) and its salts
• Removed from the list: 4,4'-Dihydroxydiphenylmethane (Bisphenol F, BPF)
• Pending status: Resorcinol has not yet been included in the official list and remains under review.
Mandatory Compliance Requirements for Enterprises (Unchanged from Previous Versions)
The update of the list does not change the compliance thresholds. Enterprises shall continue to fulfill the following obligations:
• Where the mass fraction of a substance > 0.1%: Information shall be communicated throughout the supply chain, and relevant safe-use documentation shall be provided to partners; responses to consumer inquiries shall be completed within 45 days.
• Where the mass fraction of a substance > 0.1% and annual export volume > 1 ton: A compliance notification must be submitted to the relevant EU official authorities.
One-Stop Compliance Support to Safeguard the EU Market
Relying on an experienced technical team and a full-process laboratory service system, we provide full-cycle compliance solutions covering the reach regulation, paired with customized SVHC testing services. These services precisely meet the requirements of the updated list, helping enterprises efficiently conduct ingredient screening and compliance management, and achieve a stable layout in the EU market.
Email:hello@jjrlab.com
Write your message here and send it to us
 Amazon Clothing Storage Units ASTM F2057-23 and DV
Amazon Clothing Storage Units ASTM F2057-23 and DV
 What is the ASTM F2057 Compliance Standard?
What is the ASTM F2057 Compliance Standard?
 US Dresser ASTM F2057-23 Furniture Tip Over Test
US Dresser ASTM F2057-23 Furniture Tip Over Test
 The Latest 2026 Thailand NBTC Compliance Guide
The Latest 2026 Thailand NBTC Compliance Guide
 CE Certification Directives for Electronic Product
CE Certification Directives for Electronic Product
 What is CPSC eFiling
What is CPSC eFiling
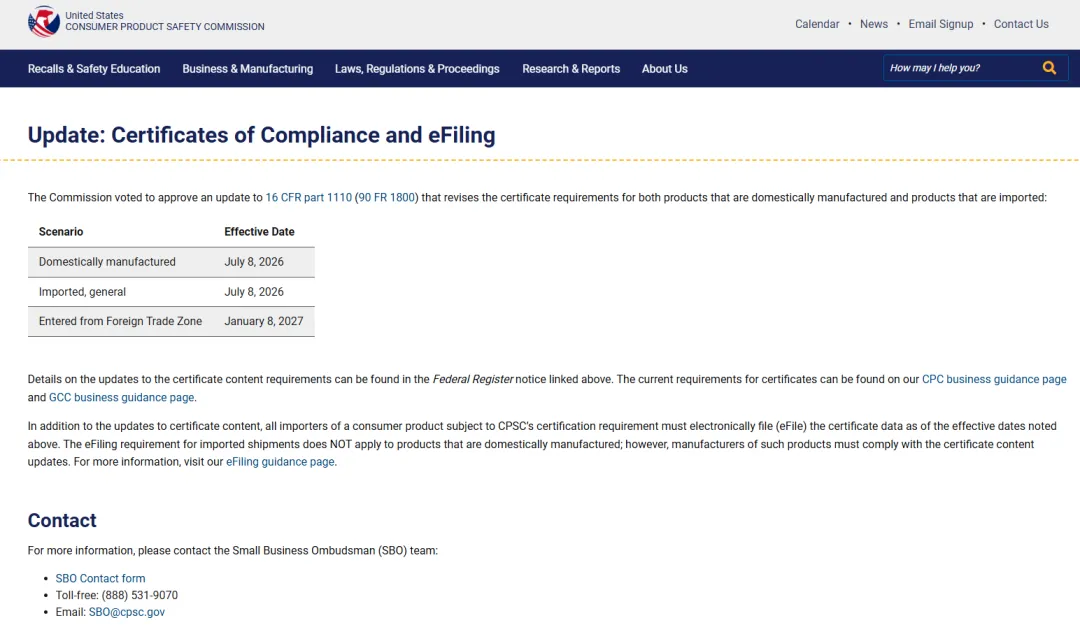 CPSC Products Must Undergo eFiling
CPSC Products Must Undergo eFiling
 What is the U.S. CPSC eFiling System?
What is the U.S. CPSC eFiling System?
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




