
Differences Between FDA and LFGB for Food Contact Materials
Food Contact Materials (FCM) refer to materials that come into direct contact with food during the normal use of products. These materials include, but are not limited to, plastics, rubber, resins, silica gel, metals, alloys, paper, cardboard, glass, ceramics, enamel, colorants, printing inks, etc. They are widely used in fields such as food packaging, tableware, kitchen utensils, food processing machinery, and kitchen electrical products.

What is FDA Registration
FDA is the abbreviation of the U.S. Food and Drug Administration, an executive agency established under the U.S. Department of Health and Human Services and the Public Health Service. Its main responsibilities include the supervision and inspection of products such as food, drugs, medical devices, food additives, cosmetics, etc.
What is LFGB Certification
lfgb certification, also known as the Law on the Regulation of Food, Tobacco Products, Cosmetics and Other Daily Necessities, is the most important basic legal document in Germany's food hygiene management system. It serves as the guideline and core for formulating other special food hygiene laws and regulations.
Differences Between FDA and LFGB
1. Regional Application Differences
FDA registration pertains to food certification in the United States, while LFGB certification applies to food-related certification in Germany. The two sets of standards follow the respective national regulations, with no overlap between them, forming completely independent systems.
2. Differences in Testing Items
Article 30 and Article 31 of Germany's LFGB stipulate the following testing items:
1. Initial inspection of samples and materials;
2. Sensory evaluation of odor and taste transfer;
3. Plastic samples: Testing of transferable components and leachable heavy metals;
4. Metals: Testing of composition and leachable heavy metals;
5. Silicone resins: Testing of transferable or volatile organic compounds;
6. Special materials, etc.
FDA registration covers different testing items as follows:
7. Overall migration test: Determining the amount of harmful substances migrating from materials to food under specific temperature and contact duration conditions that simulate real food contact scenarios;
2. Heavy metal migration test: Measuring the migration limits of heavy metals (such as lead, cadmium, chromium, arsenic, mercury, etc.) from materials to food;
3. Volatile organic compound (VOC) migration test: Testing the migration limits of volatile organic compounds from materials to food;
4. Specific substance migration test: Setting specific migration limits for certain substances, including heavy metals and volatile organic compounds.
3. Differences in Testing Content
A key feature of FDA certification is that it only measures the total amount of migrated substances, without detecting specific migrated components.
In contrast, LFGB certification not only tests the total migrated amount but also detects the migration amount of individual monomers.
4. Differences in Testing Conditions
For aqueous solution media:
① Under FDA standards, the limit specified in 21 CFR 2600 is relatively high, making it easier to pass the test, while compliance with 21 CFR 1810 is more challenging.
② LFGB testing for aqueous solutions is generally easy to pass, but the test using isooctane as the medium is comparatively difficult.
•
Notes on LFGB Certification
The key difference between Germany's LFGB and other food-grade testing standards lies in its comprehensive testing requirements, which are stricter than those of most other countries. Moreover, LFGB test results are more accurate, which is why LFGB Test Reports are widely recognized and accepted by other countries around the world.
Email:hello@jjrlab.com
Write your message here and send it to us
 Amazon Clothing Storage Units ASTM F2057-23 and DV
Amazon Clothing Storage Units ASTM F2057-23 and DV
 What is the ASTM F2057 Compliance Standard?
What is the ASTM F2057 Compliance Standard?
 US Dresser ASTM F2057-23 Furniture Tip Over Test
US Dresser ASTM F2057-23 Furniture Tip Over Test
 The Latest 2026 Thailand NBTC Compliance Guide
The Latest 2026 Thailand NBTC Compliance Guide
 CE Certification Directives for Electronic Product
CE Certification Directives for Electronic Product
 What is CPSC eFiling
What is CPSC eFiling
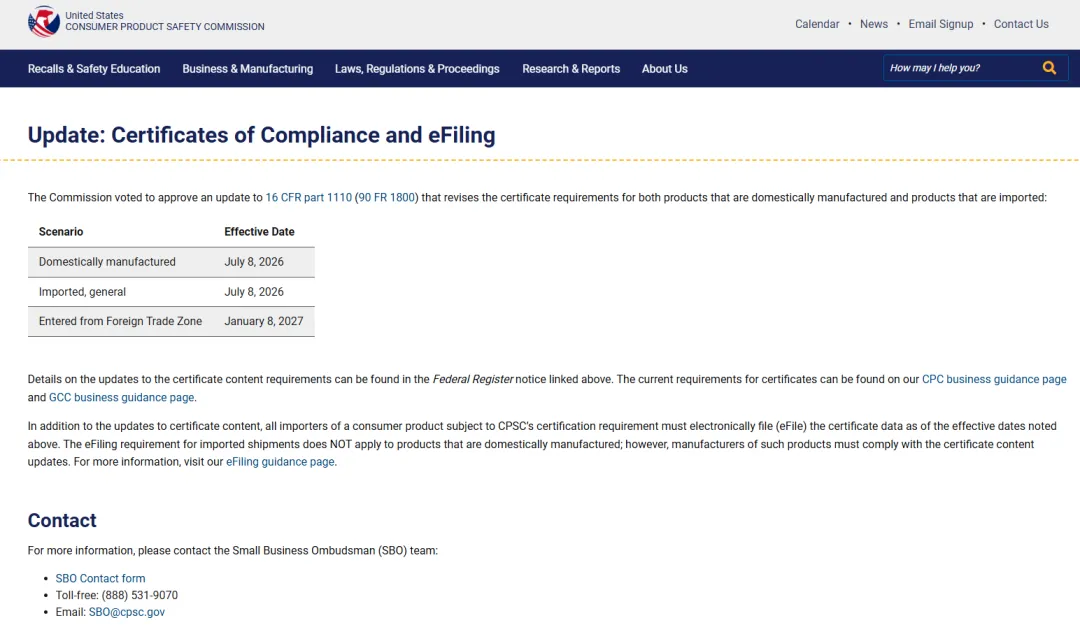 CPSC Products Must Undergo eFiling
CPSC Products Must Undergo eFiling
 What is the U.S. CPSC eFiling System?
What is the U.S. CPSC eFiling System?
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




