
UK Cosmetics Responsible Person Service
In accordance with the relevant regulations of overseas regulatory authorities, product labelling must meet the compliance requirements of the target sales market. Based on this, this document, combined with relevant requirements, aims to provide merchants with compliance references, helping them better achieve compliance in product labelling and protect the legitimate rights and interests of consumers.
Special Note: To ensure product compliance, please carefully check before shipment and confirm that all actual delivered products have complete label content affixed in line with relevant rules. If the product fails to comply, is returned, complained about, subject to regulatory accountability, or even recalled due to missing label information, you shall bear full responsibility. We urge you to attach great importance to this matter and conduct standardised operations to avoid unnecessary losses.
Key Compliance Points for Physical Cosmetics Labels
1. UK Responsible Person Information
A UK Responsible Person is a natural or legal person established in the UK, appointed by a manufacturer located outside the UK. It is responsible for ensuring product traceability in the UK market, maintaining technical documentation, assisting in incident reporting and recalls, and acting as the manufacturer’s compliance “responsible person” in the UK. Its information must be marked on imported products and relevant documents as required.

(Illustrations for reference only)

(Illustrations for reference only)
Note: The address of the UK Responsible Person must be real, valid, detailed, and capable of contacting the UK representative.
2. Country of Origin
Common expressions: Made in XXX / Country of origin: XXX

(Physical photo for reference only)
3. Net Content
The metric and imperial net content is usually displayed by weight, volume, etc., according to product characteristics.
If only a number plus a unit is printed (e.g., 50g, 50ml), no language requirement applies. If terms such as Net Contents or Net are mentioned, the language of the destination market must be used.
If the content inside the packaging is less than 5g or 5mL, or for free samples and single-use packaging products, only the quantity of products inside the package needs to be indicated, with no net content information required.

(Physical photo for reference only)
4. Expiry Date
Refers to the shelf-life expiry date of the product.
• If the shelf-life is 30 months or less, display the hourglass symbol (as shown below) + expiry date (expressed as Month/Year or Day/Month/Year).

• If the shelf-life exceeds 30 months, display the open-jar symbol, indicating the valid months after opening as xx M. For example, if valid for 6 months after opening, mark as shown below.
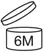

(Physical photo for reference only)
5. Special Precautions
If the product is subject to any special precautionary measures, such information shall be listed in compliance with applicable laws and regulations. A recommended format: Warning + precaution statement (note: it shall start with the word “Warning” in five languages: German, French, Italian, Spanish and English). Other regions shall still use the language of the destination market. For example,

(Examples in German, French, Italian, Spanish and English)

(Physical photo in German, French, Italian, Spanish and English, for reference only)
Note:
Warning statements and precautionary information, as well as special instructions for use, shall at least include the precautions listed in Annexes III to VI and any special precautions for professional-use cosmetics.
• ANNEX III: List of substances which cosmetic products must not contain except subject to the restrictions laid down
• ANNEX IV: List of colorants allowed in cosmetic products
• ANNEX V: List of preservatives allowed in cosmetic products
• ANNEX VI: List of UV filters allowed in cosmetic products
For more details, please visit: https://www.legislation.gov.uk/eur/2009/1223/annex/IV
6. Batch Number
Production batch number or identification number for the cosmetic product. For example,
Batch number: XXXXXX

(Physical photo for reference only)
7. Functions
Refers to the efficacy of cosmetics, usually starting with the word Functions. Efficacy information shall be provided in German, French, Italian, Spanish and English; for other countries/regions, the language of the destination market shall be used. For example,
(Examples in German, French, Italian, Spanish and English)

(Physical photo in German, French, Italian, Spanish and English, for reference only)

8. Ingredients
Refers to the product ingredient list, usually starting with the term for “ingredients” in five languages: Ingredients, Inhaltsstoffe, Ingrédients, Ingredientes, Ingredienti. The following ingredient names shall use common names, including the International Nomenclature of Cosmetic Ingredients (INCI). If no corresponding common name exists, terms from generally accepted nomenclature shall be used. For example,
Ingredients: XX, XXX


(Examples in German, French, Italian, Spanish and English)
(Physical photo in German, French, Italian, Spanish and English, for reference only)
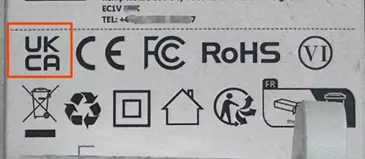
9. ukca Marking – Cosmetics
If the cosmetic product is an aerosol spray, the ukca marking must be indicated. Examples are shown below.

Or
Email:hello@jjrlab.com
Write your message here and send it to us
 RCM Certification Process & Costs for Panorami
RCM Certification Process & Costs for Panorami
 Smart Watch Australia RCM Certification
Smart Watch Australia RCM Certification
 What is RCM Certification Electronic Products?
What is RCM Certification Electronic Products?
 How much does RCM certification cost?
How much does RCM certification cost?
 CPSC eFiling Customs Clearance Guide
CPSC eFiling Customs Clearance Guide
 US CPSC eFiling Electronic Filing Compliance Guide
US CPSC eFiling Electronic Filing Compliance Guide
 2026 CPSC eFiling Process Guide
2026 CPSC eFiling Process Guide
 What is an Importer of Record
What is an Importer of Record
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




