
What is In Vitro Cytotoxicity Test?
How much does the in vitro cytotoxicity test cost?
We are a laboratory in China, and the cost of our in vitro cytotoxicity test is around $450. Our testing period is 10 working days, with an affordable price.

What is in vitro cytotoxicity testing?
In vitro cytotoxicity testing is a test using cell culture techniques, which can be used to determine cell death (e.g., cell lysis), inhibition of cell growth, colony formation, and other effects on cells caused by medical devices, materials, or their extracts.

In the biological evaluation system of medical devices, in vitro cytotoxicity testing is one of the most important test indicators. Through cytotoxicity testing, the potential in vitro cytotoxicity of medical devices and biomaterials can be evaluated. Before being used in tissues and cells of living organisms, in vitro cytotoxicity testing serves as a simulated experiment to predict reaction effects. In vitro cytotoxicity testing methods are economical, simple, and can be used for batch testing simultaneously, providing important screening materials for whether samples should undergo animal laboratory experiments.
vitro cytotoxicity testing lab
Our laboratory has many years of experience in cytotoxicity testing and has a perfect testing system for cytotoxicity testing of medical devices and biomaterials. For cytotoxicity testing of medical devices and biomaterials, testing methods can generally be divided into two categories: cytotoxicity quantitative testing and cytotoxicity qualitative testing.

Cytotoxicity Quantitative Testing: MTT Test
Principle: This test is based on the observation of cell metabolic activity. MTT is a yellow water-soluble reagent, which is reduced by living cells to form a blue-purple formazan. After dissolving the formazan in isopropanol, the optical density is measured using a spectrophotometer, and the number of viable cells is proportional to the optical density.
Reference Standards:
- iso 10993-5:2009 Biological evaluation of medical devices – Part 5: Tests for in vitro cytotoxicity.
- ISO 10993-12:2021 Biological evaluation of medical devices – Part 12: Sample preparation and reference materials.
- GB/T 16886.5-2017 Biological evaluation of medical devices – Part 5: In vitro cytotoxicity tests.
Notes: The laboratory test sampling method generally uses extraction solution for experimentation, selects appropriate extraction ratios based on the characteristics and thickness of the samples according to the standard, and further shears the samples.

Cytotoxicity Qualitative Testing: ISO Qualitative Method
Principle: Medical device leachates are cultured with cells, and the biological response of the cells is observed under a microscope. The changes in cells are evaluated from aspects such as cell morphology, vacuole formation, cell detachment, cell lysis, and cell membrane integrity, and are rated accordingly.
Reference Standards:
- iso 10993-5:2009 Biological evaluation of medical devices – Part 5: Tests for in vitro cytotoxicity.
- GB/T 16886.5-2017 Biological evaluation of medical devices – Part 5: In vitro cytotoxicity tests.

Cytotoxicity Qualitative Testing: USP Qualitative Method
Principle: This test method evaluates the effect of leachates from elastomeric or polymeric materials in direct or indirect contact with patients on cell morphology to determine the biological response of the extract.
Reference Standards: USP-NF <87>: BIOLOGICAL REACTIVITY TESTS, IN VITRO: Elution Test.
Email:hello@jjrlab.com
Write your message here and send it to us
 Amazon Clothing Storage Units ASTM F2057-23 and DV
Amazon Clothing Storage Units ASTM F2057-23 and DV
 What is the ASTM F2057 Compliance Standard?
What is the ASTM F2057 Compliance Standard?
 US Dresser ASTM F2057-23 Furniture Tip Over Test
US Dresser ASTM F2057-23 Furniture Tip Over Test
 The Latest 2026 Thailand NBTC Compliance Guide
The Latest 2026 Thailand NBTC Compliance Guide
 CE Certification Directives for Electronic Product
CE Certification Directives for Electronic Product
 What is CPSC eFiling
What is CPSC eFiling
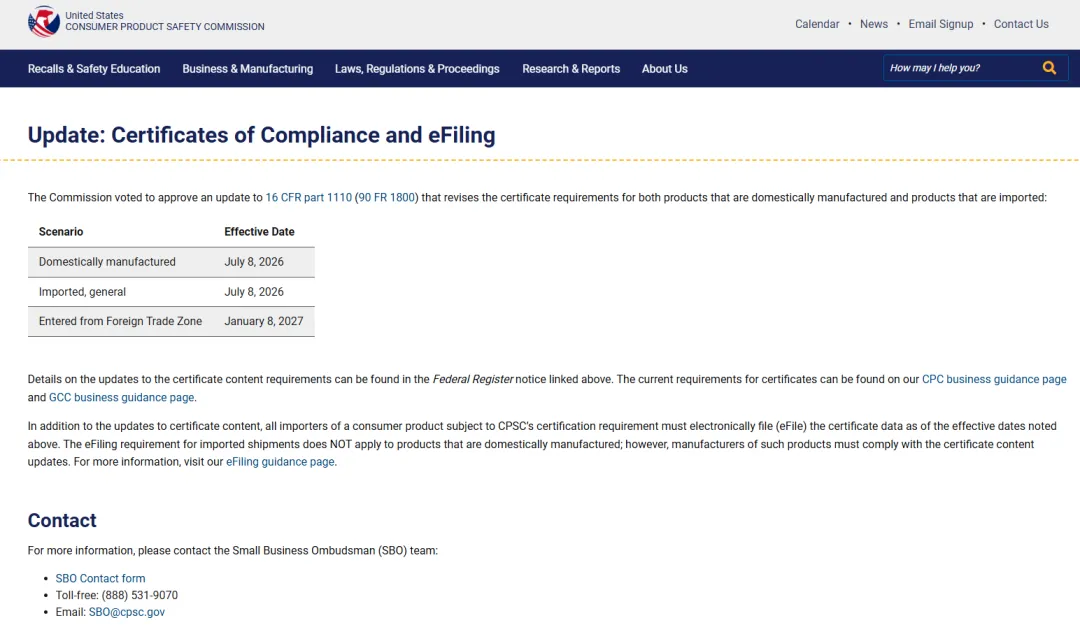 CPSC Products Must Undergo eFiling
CPSC Products Must Undergo eFiling
 What is the U.S. CPSC eFiling System?
What is the U.S. CPSC eFiling System?
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




