
Button Cell Battery PAS 7055:2021 Certification Testing
What Regulations Apply to Button Cell Batteries in the UK?
In April 2021, the British Standards Institution (BSI) published the Button/Coin Battery Safety Standard (PAS 7055:2021), which sets safety standards for button (non-lithium) batteries and coin (lithium) batteries.
This standard was developed in accordance with the UK General Product Safety Regulations 2005 (UK gpsr). Retailers selling button batteries can refer to the PAS 7055:2021 standard for compliance.
The UK’s button battery legislation is similar to the U.S. battery regulations. It mandates that consumer products using button/coin batteries—including but not limited to flashlights, electronic scales, and remote controls—must comply with the relevant safety standards.
In 2023, eBay issued related policies stating that UK regulatory authorities consider PAS 7055:2021 safety requirements when assessing product safety. The UK Office for Product Safety and Standards (OPSS) encourages businesses to implement PAS 7055:2021 battery regulations.
Similarly, TEMU has stated that it reserves the right to take action against non-compliant products, including but not limited to: responding proactively to regulatory requirements, issuing risk warnings to buyers, and removing listings.
What Are the Labeling Requirements for Button Batteries in the UK?
1) Labeling Requirements for Coin (Lithium) Battery Packaging
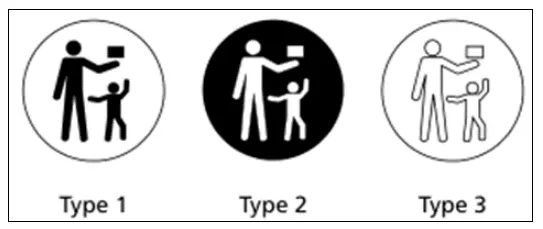
Coin or lithium battery packaging must feature a warning symbol, which means:
"Keep out of reach of children to prevent accidental ingestion." (See Figure 1)

If the symbol cannot be printed in color, a black-and-white version may be used instead (See Figure 2).
2) Labeling Requirements for Button (Non-Lithium) Battery Packaging
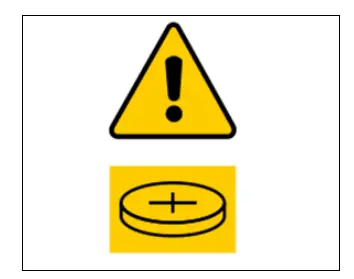
A warning label must be placed on the packaging of button or non-lithium batteries.
3) Safety Marking for Products Containing Button/Coin Batteries
Products containing button/coin batteries must display a safety marking on the product itself, preferably near or on the battery compartment. The safety marking should be:
- Permanent,
- Clearly legible, and
- Indelible.
Important: If the marking cannot be added to the product itself or if doing so would compromise product recognition, then a label with the required safety warning must be added to the packaging.
Safety Marking Requirements:
- a) Must be placed on a contrasting background, covering at least 50% of the symbol area.
- b) The diameter of the warning symbol must be at least 6mm.
- c) The text "BATTERY WARNING: KEEP OUT OF REACH OF CHILDREN" must contrast with the background color.

What Are the Warning Label Requirements for Button/Coin Batteries and Products Containing Them?
1) Button/Coin Battery Warning Labels:
- "BATTERY WARNING: KEEP OUT OF REACH OF CHILDREN"
- "If you think batteries might have been swallowed or placed inside any part of the body, seek immediate medical attention."
2) Warning Labels for Packaging of Products Containing Button/Coin Batteries:
- "BATTERY WARNING: KEEP OUT OF REACH OF CHILDREN"
- "If the battery compartment (if applicable) does not close securely, stop using the product and keep it away from children."
- "If you think batteries might have been swallowed or placed inside any part of the body, seek immediate medical attention."
It is important to note that all warning labels on packaging must be permanent, clearly legible, and indelible. Additionally, the warning must remain intact and undistorted when batteries are removed, such as when taken out from a multi-pack.
If you have any further questions regarding button battery certification in the UK or the US, feel free to contact us!
Email:hello@jjrlab.com
Write your message here and send it to us
 What Are the Safety Tests for Lithium Batteries?
What Are the Safety Tests for Lithium Batteries?
 Wireless Device EMC Certification
Wireless Device EMC Certification
 Type-C Interface EN62680-1 Testing
Type-C Interface EN62680-1 Testing
 Electrical Safety Test IEC 60601
Electrical Safety Test IEC 60601
 IEC 60601-1-2 EMC Testing
IEC 60601-1-2 EMC Testing
 Polycyclic Aromatic Hydrocarbons (PAHs) Testing La
Polycyclic Aromatic Hydrocarbons (PAHs) Testing La
 EU EN71 Certification Testing for Mechanical Toys
EU EN71 Certification Testing for Mechanical Toys
 EU Toy Safety Testing (EN 71 / 2009/48/EC)
EU Toy Safety Testing (EN 71 / 2009/48/EC)
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




