
How Much Does a Cytotoxicity Test Cost
Introduction to cytotoxicity test
Cytotoxicity test is an in vitro testing method that simulates the growth environment of organisms in an isolated state to detect the biological responses induced by medical devices, biomaterials or chemicals after contacting body tissues.
Cytotoxicity itself refers to a simple cell-killing event caused by cells or chemicals, which does not depend on the specific cell death mechanisms of apoptosis or necrosis. When cells are exposed to toxic substances, they may exhibit a series of responses such as necrosis (uncontrolled cell death), apoptosis (programmed cell death), autophagy, or cessation of growth and division.

In the field of medical devices and biomaterials, cytotoxicity testing plays a vital role. Since these products will ultimately be used in the human body and directly contact human tissues and cells, they are required to possess not only excellent physical and chemical properties but also reliable biocompatibility. As one of the most critical indicators in the biocompatibility evaluation system, cytotoxicity testing is designated as a mandatory item for the biological evaluation of medical devices, thanks to its advantages of simplicity, rapidity, high sensitivity and reduced reliance on laboratory animals.
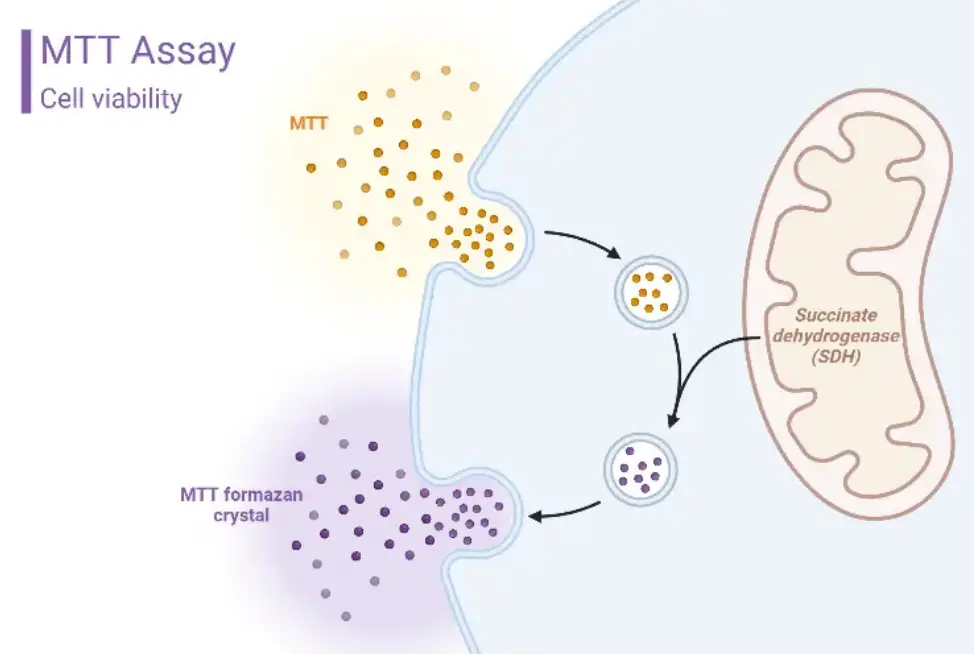
The degree of cytotoxicity is mainly evaluated by detecting changes in cell membrane permeability. Common testing methods include the MTT assay, CCK-8 assay, LDH release assay, agar diffusion method and direct contact method.
Standards for Cytotoxicity Test
The ISO 10993 series standards constitute the core standard system for the biological evaluation of medical devices, among which iso 10993-5 is specifically dedicated to in vitro cytotoxicity testing. This standard specifies the basic requirements and method selection criteria for in vitro cytotoxicity testing of medical devices, and is widely recognized by international regulatory authorities such as the U.S. FDA and the EU CE certification.
Cost of Cytotoxicity Test
① Price at JJR Laboratory: $700
② Sample Submission & Consultation Welcome
You can contact us via email and send your product specifications. We will issue a commercial invoice containing details such as testing fees, applicable standards and testing cycle.
As a key method for evaluating the biological safety of medical devices, pharmaceuticals and cosmetics, the cost of cytotoxicity testing is affected by multiple factors, ranging from several hundred to several thousand US dollars for a comprehensive assessment. For specific inquiries, please feel free to contact us.
Email:hello@jjrlab.com
Write your message here and send it to us
 How to Perform a Valid CCK-8 Cytotoxicity Assay
How to Perform a Valid CCK-8 Cytotoxicity Assay
 Introduction to Cytotoxicity Assay (CCK-8 Method)
Introduction to Cytotoxicity Assay (CCK-8 Method)
 What Are the Cytotoxicity Assay Methods?
What Are the Cytotoxicity Assay Methods?
 What is Ingress Protection (IP) Testing and Standa
What is Ingress Protection (IP) Testing and Standa
 What is UL 153 Lighting Standards and Portable Lum
What is UL 153 Lighting Standards and Portable Lum
 What is Conducted and Radiated Emissions Testing?
What is Conducted and Radiated Emissions Testing?
 What is FCC SDoC Certification Compliance
What is FCC SDoC Certification Compliance
 What Are RoHS, UN 38.3 and MSDS, and How Do They D
What Are RoHS, UN 38.3 and MSDS, and How Do They D
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




