
What is ISO 10993 Biocompatibility Testing?
For any medical device intended for the global market, ISO 10993 biocompatibility testing is the first and critical scientific proof of its safety and performance.
Biological safety means that when materials interact with the human body, they must be non-toxic, non-sensitizing, non-irritating, non-genotoxic, non-carcinogenic, and cause no adverse reactions to human tissues, blood, or the immune system.
JJR Medical & Biological Laboratory has obtained China CMA and U.S. IAS accreditations. The laboratory strictly operates in accordance with the GLP system and is recognized by regulatory authorities including the U.S. FDA, Australia TGA, and Brazil ANVISA.
It is also an accredited laboratory and partner of many international organizations, such as UL (USA), CSA (Canada), TÜV (Germany), SGS (Switzerland), Intertek (UK), BSI (UK), MEDCERT (Germany), and IMQ (Italy).
JJR Laboratory provides professional, efficient, and comprehensive biocompatibility evaluation and biocompatibility testing report services.

Biocompatibility Testing Items
Test Item | Reference Standard | Testing Cycle |
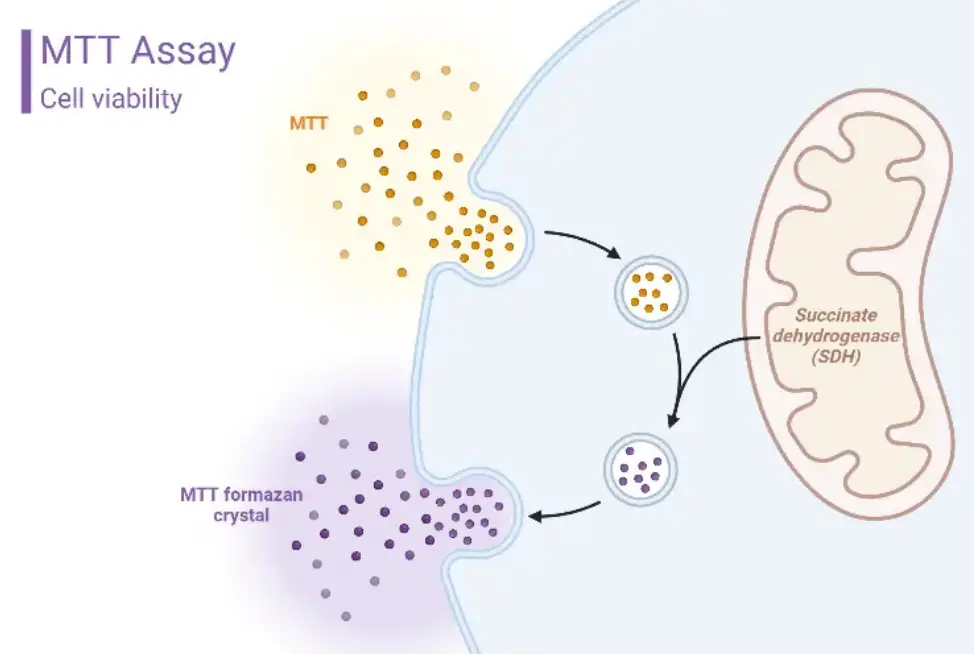
Cytotoxicity Test (MTT Assay) | iso 10993-5 / USP 87 / GB/T 16886.5 / ISO 7405 / YY 0127.9 / GB/T 14233.2 | 2 weeks |
Cytotoxicity Test (Agar Overlay Method) | 2 weeks | |
Cytotoxicity Test (Filter Diffusion Method) | 2 weeks | |
Cytotoxicity Test (Direct Contact Method) | 2 weeks | |
Cytotoxicity Test (Elution Method) | 2 weeks | |
Irritation and Sensitization Tests | ||
Sensitization Test (Maximization / Patch Test) | iso 10993-10 / GB/T 16886.10 / GB/T 14233.2 | 6–8 weeks |
Skin Irritation Test | ISO 10993-10 / GB/T 16886.10 | 2 weeks |
Intradermal Irritation | ISO 10993-10 / GB/T 16886.10 / USP 88 / GB/T 14233.2 | 2 weeks |
Oral Irritation (histopathology required) | ISO 10993-10 / GB/T 16886.10 | 6–8 weeks |
Vaginal Irritation (histopathology required) | 6–8 weeks | |
Penile Irritation (histopathology required) | 6–8 weeks | |
Rectal Irritation (histopathology required) | 6–8 weeks | |
Eye Irritation | 4 weeks | |
Systemic Toxicity Tests | ||
Acute Systemic Toxicity Test (IV / Oral) | ISO 10993-11 / GB/T 16886.11 / USP 88 / GB/T 14233.2 | 2 weeks |
Subacute Systemic Toxicity (14d / 28d, no histopathology) | ISO 10993-11 / GB/T 16886.11 / GB/T 14233.2 | 4 / 6 weeks |
Subacute Systemic Toxicity (14d / 28d, with histopathology) | ISO 10993-11 / GB/T 16886.11 / GB/T 14233.2 | 6 / 6–8 weeks |
Subchronic Systemic Toxicity (90d) | 4–4.5 months | |
Chronic Systemic Toxicity (180d) | 7.5–8 months | |
Pyrogen Test | ISO 10993-11 / GB/T 16886.11 / Chinese Pharmacopoeia | 2 weeks |
Implantation Tests | ||
Subcutaneous Implantation (histopathology required) – Rabbit, Rat | iso 10993-6 / GB/T 16886.6 / USP 88 | – |
Muscle Implantation (histopathology required) – Rabbit, Rat | ISO 10993-6 / GB/T 16886.6 / USP 88 | – |
Bone Implantation (histopathology required) – Rabbit, Rat | ISO 10993-6 / GB/T 16886.6 | – |
Hemocompatibility Tests | ||
Hemolysis Test (rabbit blood preferred) | ISO 10993-4 / GB/T 16886.4 / GB/T 14233.2 | 2 weeks |
Hemolysis Test (rabbit blood preferred) | astm f756 (customer-specified) | 4–6 weeks |
Coagulation | ISO 10993-4 / GB/T 16886.4 | 4–6 weeks |
Platelet Count | 4–6 weeks | |
Total Complement | 4–6 weeks | |
Thrombosis (in vivo canine CTI / in vitro) | 4–6 weeks | |
Genotoxicity Tests | ||
Ames Salmonella Typhimurium Reverse Mutation Assay | ISO 10993-3 / GB/T 16886.3 / YY/T 0127.10 | 6–8 weeks |
Gene Mutation Test | ISO 10993-3 / GB/T 16886.3 / OECD 476 | 6–8 weeks |
Chromosomal Aberration Test | ISO 10993-3 / GB/T 16886.3 / OECD 473 | 6–8 weeks |
Sperm Abnormality Test (rat, microscopic reading) | ISO 10993-3 / GB/T 16886.3 | 6–8 weeks |
Micronucleus Test (mouse, microscopic reading) | ISO 10993-3 / GB/T 16886.3 / OECD 474 | 6–8 weeks |
What is the ISO 10993 Standard Series?
The ISO 10993 series, titled Biological Evaluation of Medical Devices, is a globally recognized authoritative standard issued by the International Organization for Standardization.
Its core purpose is to assess potential biological risks of medical device materials when in contact with the human body, ensuring acceptable biocompatibility — meaning materials do not cause unacceptable adverse reactions in their intended application.
The series includes more than 20 parts, covering risk management, sample preparation, specific testing (cytotoxicity, sensitization, irritation, systemic toxicity, etc.), and data interpretation.
ISO 10993-1: Evaluation and testing in risk management processes acts as the framework document that establishes a risk-based assessment strategy.
For more information about biocompatibility testing and evaluation, please contact JJR Medical & Biological Laboratory.
Test reports issued by JJR Laboratory are recognized by major EU certification bodies (TÜV, SGS, BSI, IMQ, MEDCERT, etc.) and authorities including the U.S. FDA, Australia TGA, Canada CSA, Nigeria SONCAP, and NAFDAC.
Email:hello@jjrlab.com
Write your message here and send it to us
 ISO 10993-10 In Vitro Skin Sensitization Test Serv
ISO 10993-10 In Vitro Skin Sensitization Test Serv
 What is ISO 10993 Biocompatibility Testing?
What is ISO 10993 Biocompatibility Testing?
 How to Perform a Valid CCK-8 Cytotoxicity Assay
How to Perform a Valid CCK-8 Cytotoxicity Assay
 Introduction to Cytotoxicity Assay (CCK-8 Method)
Introduction to Cytotoxicity Assay (CCK-8 Method)
 What Are the Cytotoxicity Assay Methods?
What Are the Cytotoxicity Assay Methods?
 What is Ingress Protection (IP) Testing and Standa
What is Ingress Protection (IP) Testing and Standa
 What is UL 153 Lighting Standards and Portable Lum
What is UL 153 Lighting Standards and Portable Lum
 What is Conducted and Radiated Emissions Testing?
What is Conducted and Radiated Emissions Testing?
Leave us a message
24-hour online customer service at any time to respond, so that you worry!




